Back to Journals » Open Access Emergency Medicine » Volume 16
The Role of Ophthalmology in Tele-Stroke Consults for Triaging Acute Vision Loss
Authors Artymowicz A, Douglas C, Cockerham K
Received 11 December 2022
Accepted for publication 18 September 2023
Published 5 February 2024 Volume 2024:16 Pages 45—56
DOI https://doi.org/10.2147/OAEM.S395588
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Hans-Christoph Pape
Anna Artymowicz,1 Christina Douglas,2 Kimberly Cockerham2,3
1West Coast Eye Institute, Bakersfield, CA, USA; 2Southeast Eye Specialists, Knoxville, TN, USA; 3Senta Clinic, San Diego, CA, USA
Correspondence: Anna Artymowicz, West Coast Eye Institute, 215 China Grade Loop, Bakersfield, CA, 93308, USA, Tel +1 (661) 393-2331, Email [email protected]
Abstract: Advances in telemedicine have allowed physicians to provide care in areas that were previously geographically or practically inaccessible. Roughly 70% of all US hospital have less than 200 bed capacity and nearly 50% have fewer than 100 beds. These smaller hospitals often do not have specialists available for bedside patient care, making them potential beneficiaries of telemedicine medical specialty services. In 2005, the American Stroke Association proposed implementing telemedicine services in effort to increase access to acute stroke care in neurologically underserved areas such as small hospitals. Tele-stroke services have since become established across the country and are now utilized by approximately 30% of US hospitals. By reducing the time between presentation and evaluation by a stroke specialist, tele-stroke programs have successfully increased patient access to life-saving treatment with tissue-plasminogen activator (t-PA) treatments. This change has been especially profound remote and underserved community hospitals. However in the evaluation of acute vision loss, an area where ophthalmology and stroke care overlap, increased reliance on tele-stroke services has contributed to some unique challenges. Acute vision has a complex differential and is commonly a result of conditions other than stroke. When tele-stroke services are engaged for the evaluation of acute vision loss, the neurologist is asked to make medical decisions without complete information about the eye. This situation can expose patients to costly or inappropriate testing, unnecessary hospitalizations, or lead to delayed diagnosis and treatment of non-neurologic conditions of the eye. The goal of this paper is to provide an overview of the overlap between stroke and vision loss, highlight the challenges inherent in using tele-stroke in evaluating acute vision loss and to offer our comments on how increased communication between emergency medicine, ophthalmology, and neurology services can ensure that patients with vision loss receive the highest standard of care in all hospitals.
Keywords: telemedicine, tele-stroke, acute vision loss, tele-neurology, CRAO
Introduction
Visual function is determined by the health and relationship between the periocular structures, the globe, and the visual pathways in the brain. A disruption in any of these components can lead to changes or decrease in vision. Of the many etiologies that can lead to acute vision loss, some require a full ophthalmic exam for diagnosis and management, others require a neurologic evaluation, and some may require both (Figure 1).
Tele-stroke services and are utilized by approximately 30% of US hospitals.1 As ophthalmologists, we have noticed an increasing incidence of people presenting to emergency departments with acute vision loss who are assessed by tele-stroke services, receive a full stroke workup, and are later diagnosed with conditions that could have been diagnosed on ophthalmic exam. In these cases, a timely ophthalmic exam could have prevented unnecessary, costly, or invasive testing, as well as often eliminating the need for a hospital admission. We postulate that the increased reliance on tele-stoke services without a simultaneous increase in accessibility of ophthalmic specialty services contribute to this.
The window for treating eligible patients in cases of ischemic stroke with tissue plasminogen activator (tPA) is within 3–4.5 hours of the onset of symptoms.2,3 Therefore, when a patient presents to the emergency department with signs of possible acute cerebral stroke, the priority is acting fast to rule in or out the stroke diagnosis. In the case of acute vision loss, a possible symptom of cerebral stroke or a retinal arterial occlusion, an emergent ophthalmic exam can rule in or out many of the non-neurologic causes for the vision change (see Figure 1). Below, we review the overlap between stroke and visual loss. We will then discuss challenges inherent in both in-person and tele-medicine ophthalmic exams and end with comments on how a multi-specialty approach incorporating tele-ophthalmic technologies into tele-stroke protocols can help us provide the highest quality of care for all patients regardless of resource status of the hospital.
Stroke and Vision Loss – Current Management and Implications
Our aim in the following section is to not to provide a comprehensive review of stroke management but instead to focus on recommendations for those in whom an arterial occlusion is the suspected etiology of vision loss in the context of increasing availability of and reliance on tele-stroke programs.
Cerebral Ischemia and Vision Loss
The spectrum of visual dysfunction resulting from cerebral stroke is varied and large. Ischemia of the lateral geniculate body (LGB), optic radiations, or occipital lobe can manifest as homonymous or incongruous quadrantinopias, sectoranopias or scotomas.4 Occipital pole ischemia can cause a complete hemianopia, whereas bilateral posterior infarctions can result in total blindness without the patient recognition of the loss, called Anton’s Syndrome. Midbrain infarctions, on the other hand, can result in ptosis or ophthalmoplegia due to cranial nerve palsies.
Notably, strokes are not the only cerebral causes for visual loss or change. Other causes include demyelinating disease, mass effects, infectious and inflammatory etiologies, toxicities, metabolic disturbances such as hepatic encephalopathy, seizures and migraines.5 These cerebral causes for vision changes should be considered, especially when the vision change is bilateral.
It is important to note that though vision loss can be the only presenting symptom of cerebral stroke,6 it is exceedingly rare for stroke-related vision loss to be present without other stroke symptoms. Even in conjunction with other symptoms, vision changes are one of the presenting symptoms in only 24–26% of stroke patients.7,8 Interestingly, even though 68–92% of patients diagnosed with an acute stroke are subsequently found to have an associated visual impairment, only 40% of multidisciplinary stroke teams report objective findings of ocular impairment during the emergent care9 and at least equally many patients remain unaware of their vision deficits even after formal visual testing.10,11 The discrepancy between the vision changes reported by the patients and their physicians in the emergency setting and the later objective vision findings is likely multifactorial, including subjective difficulty in identifying incongruous or small visual field deficits and the low sensitivity of confrontational visual field testing performed in the emergency setting.12
Implications
Vision change is rarely the sole presenting symptom of a cerebral stroke. If a patient presents with vision changes in concurrence with other symptoms or signs consistent with stroke, they should be triaged, evaluated and managed like any other stroke suspect. Patients with acute and painless vision changes should be assessed with confrontational visual field testing for pathognomonic cerebral ischemic patterns of visual loss such as homonymous hemianopia. If visual field defects are not found and the patient has no other signs or symptoms that suspicious for cerebral stroke or CRAO, the patient can be referred for outpatient management by ophthalmology, preferably within 24 hours of discharge. Patients diagnosed with acute cerebral stroke with concurrent vision changes can similarly be referred to outpatient ophthalmology for a full ophthalmic exam and serial visual field tests after receiving acute stroke care and discharge from the hospital.
Retinal Artery Occlusions
Retinal ischemia can result from an occlusion of the central retinal artery (CRAO), a retinal artery branch (branch retinal artery occlusion, BRAO), the posterior ciliary artery, or rarely due to an occlusion of the ophthalmic artery. An acute CRAO can often be identified on fundoscopic exam by the classic finding of retinal whitening with a cherry red foveal spot. However, this sign classically fades over a period of weeks, and prior retinal ischemic events may have subtle fundus findings at best. Moreover, the retinal lipid accumulation in Tay-Sachs disease or macular infarction from trauma or diabetic retinopathy can also present with a cherry-red spot without the presence of arterial occlusion.
Retinal artery occlusions are more common in elderly patients but remain overall rare, estimated to have the annual incidence of around 2 per 100000 persons.13 It is estimated that 92–95% of retinal artery occlusions occur due to thromboembolic disease, while 5–8% occur in association with vasculitis, typically giant cell arteritis (GCA).14,15
Retinal ischemia can be transient, with resolution of the occluding embolus before permanent ischemic damage occurs such that the symptoms and signs of ischemia resolve by the time the patient is evaluated. Transient vision loss (TVL) due to temporary retinal arterial occlusion (also called amaurosis fugax) is almost exclusively monocular, with an estimated incidence of 1.5/100,00 people per year in the third decade of life, increasing to 32/100,000 in the seventh decade of life.16 Binocular transient vision loss due to arterial occlusions is rare and is almost always associated with severe occlusive disease of the internal carotid artery (ICA), aortic arch, or vertebral circulation with occipital lobe ischemia. In patients younger than 45 years old, transient monocular vision loss (TMVL) is more likely to be secondary to vasospasm or migraine as opposed to arterial occlusions.
Recognition and treatment of the underlying vasculopathic risk factors in patients with permanent or transient retinal ischemia can significantly reduce a patient’s risk for future stroke. Emergent diagnosis and treatment for the underlying cause acute retinal ischemia can decrease the risk for subsequent stroke by 80%.17
The European Assessment Group for Lysis in the Eye (EAGLE) study found that 40% of their patients with CRAO had ipsilateral carotid artery stenosis of 70% or greater.18 Of those experiencing transient monocular vision loss due to retinal artery occlusions, ~10 to 15% develop a new stroke within 90 days, half of which occur within the first 48 hours.19 Because those with acute retinal artery occlusions are at a higher risk for subsequent cerebral strokes, the American Heart Association/American Stroke Association (AHA/ASA) recommends emergent neuroimaging for patients with confirmed or highly suspected retinal arterial occlusions.20 MRI scanning has demonstrated evidence of cerebral ischemia in up to 4% of patients with transient monocular vision loss due to transient retinal ischemia, 25%-40 of those with branch retinal artery occlusions and 30–33% with central retinal artery occlusions.21,22
Treatment of retinal artery occlusions is controversial and traditionally understood to be unsuccessful. Conservative treatment modalities include ocular massage, inhalation of carbogen, hyperbaric oxygen, anterior chamber needle decompressions, and pharmacologic reduction of intraocular pressure, all thought to increase chances of clot dislodgement and reinstatement of normal arterial flow. The benefit of these treatments is mostly theoretical, cited in case reports with mixed outcomes.23
Though it is not the standard of care at this time, there is growing interest in treating acute central retinal arterial occlusions with intravascular thrombolysis therapies. In 2010, the European Assessment Group for Lysis in the Eye (EAGLE) randomized controlled trial found that treating CRAO in people with symptoms for 20 hours or less with tPA did not improve visual outcomes and was associated with a significantly increased risk of symptomatic intracranial hemorrhage.24 However, a 2020 meta-analysis that incorporated newer studies demonstrated a strong effect with treatment of CRAO with intravenous tPA within 4.5 hours of onset.25 In light of this, in 2021 the American Heart Association/American Stroke Association (AHA/ASA) published a scientific statement emphasizing that treatment with IV tPA may be effective for CRAO if administered in the same time-window that applies for cerebral ischemic strokes.14 Thus currently, treatment with intra-arterial tPA may be considered if patients with CRAO without contraindications for IV tPA present to specialized centers within 4–6 hours of visual loss.
Implications
If suspicion for CRAO or transient monocular vision loss from temporary retinal arterial occlusion is high, consider emergent neuroimaging based on AHA/ASA recommendations. In those patients with microvascular or hypercoagulable risk factors, an episode of TMVL should trigger an investigation for thromboembolic disease or hypercoagulable state. If a patient’s transient vision changes have resolved, the patient’s vision and ocular exam can be followed on an outpatient basis by ophthalmology.
If suspicion for CRAO or TMVL from arterial occlusion is high in a patient above the age of 50, we recommend obtaining a CBC, ESR, CRP, as well as taking a thorough history to assess for clinical symptoms of GCA (Box 1). When the clinical suspicion for GCA is high, immediate treatment with high dose IV steroids is indicated to prevent contralateral visual loss as well as prevent further morbidity, followed by a temporal artery biopsy within two weeks of starting treatment. Missing the diagnosis of a vasculitis is a common error in this population and if left untreated, can result in many systemic, neurologic, and ophthalmologic complications.
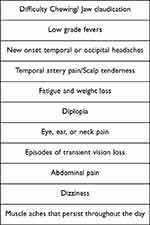 |
Box 1 Common Giant Cell Arteritis Symptoms |
In emergency centers with in-person neurologic and ophthalmologic specialty coverage, a patient with isolated acute onset painless monocular loss would receive an emergent ophthalmologic examination. In settings without access to emergent in-person or telemedicine-assisted ophthalmologic examinations, our recommendation is to perform an ultrasound of the eye to look for evidence of vitreous opacities or retinal detachment that could explain the vision loss. If the patient is younger than 50 years of age, the pupillary and the external examination of the eye is unremarkable, and the patient has no vasculopathic risk factors and exhibits no signs or symptoms suspicious for stroke, they should be considered for an outpatient ophthalmologic evaluation.
Challenges of the Ophthalmic Exam in the Emergency Setting
In-Person Emergency Ophthalmic Examination
A funduscopic exam can rule in or out numerous causes of acute, painless vision loss, including vitreous hemorrhage, intraretinal hemorrhage, retinal detachment, advanced glaucoma, venous or arterial occlusions, and macular changes from diseases such as age-related macular degeneration, polypoidal choroidal vasculopathy, or central serous choroidal retinopathy (Figure 1.)
In tertiary care centers, stroke specialists can rely on information gained from an expert ophthalmologic exam when deciding whether to pursue stroke protocols, treatment, or hospital admission for patients with acute vision loss. The question emerges – why are not fundus examinations a routine part of the emergency room exam in patients who present with acute vision loss? The challenge in obtaining a fundus exam in emergency departments is multifold – it requires specialty equipment, significant technical skill, and the knowledge to interpret the findings. Few medical professionals feel comfortable with performing a fundoscopic examination, as the examination technique is not included in the standard medical school curriculum and can take years to master. Ophthalmology has not been a required element in medical schools for over two decades. Though ED providers and neurologists may be taught how to perform basic fundoscopic exams in their residency training programs, in reality these exams are rarely performed by non-ophthalmologists. One study found that of 350 patients who presented to the emergency department with signs and symptoms warranting an examination of the ocular fundus, only 14% had a funduscopic examination performed by an emergency physician.26 Of these patients, 12.6% were later found to have fundus findings that would have changed the course of management in the ED if they were noted at the time. Notably, none of these findings were detected on the funduscopic examination by the emergency physicians. In the writers’ experience, this study reflects of the reality of the field - The ability to perform and interpret the fundoscopic exam remains something outside of the scope of the majority of providers who are not trained in ophthalmology or optometry.
The challenge of obtaining a full ophthalmic exam in the emergency setting is compounded by the growing demand for ophthalmologists for the aging population. In 2016, the Health Resources and Services Administration estimated that the number of available ophthalmologists would fall short of the demand by more than 6000 physicians by 2025.27 Roughly 70% of all US hospital have less than 200 bed capacity and nearly 50% have fewer than 100 beds.28 Ophthalmologists are rarely on staff in small hospitals, and much less available for in-house emergency evaluations. In 104 of surveyed facilities in Florida, nonrural hospitals reported a median of 1 ophthalmologist available on call compared with a median of 0 at rural hospitals.29 In California, only 49% of rural facilities and 74% of nonrural facilities30 reported having emergency ophthalmology coverage. In both states, 50–73% of the facilities or practitioners surveyed rated teleophthalmology as having high or very high value.29
Telemedicine Emergency Ophthalmic Examination
The development of new technologies to obtain and analyze many parts of the ophthalmic examination is changing the landscape of ophthalmology and the way ophthalmologists can participate in multi-disciplinary teams to triage patients in the emergency room setting. Various tools have been developed to enable non-ocular specialists to acquire critical information about the ocular system and facilitate remote evaluations by ocular specialists (Table 1). These developments have the potential to reduce the reliance on in-person ophthalmic specialty care, which is particularly important in rural and resource poor healthcare settings.
 |
Table 1 Teleophthalmologic Tools in Published Literature |
Multiple studies have shown that by sending digital images of ophthalmic studies to remotely located ophthalmologists, tele-ophthalmology can be used to diagnose patients with retinal disease, glaucoma, and other conditions usually managed in the non-emergent setting.54 Teleophthalmology services used for emergency triaging also show promise. A prospective, observational study of 129 patients presenting to the Massachusetts Eye and Ear Infirmary emergency ward compared the diagnosis and urgency designation of ophthalmologists completing remote assessment to the gold standard in-person assessment. The study found a positive predictive value of an urgent telemedical triage of 0.93 (95% CI 0.88, 0.99) and a negative predictive value of a non-urgent telemedical triage of 0.67 (95% CI 0.56, 0.77).55
However, current integration of teleophthalmology into telestroke protocols for assessment of acute vision loss remains suboptimal. One review article found that of the 90 discrete teleophthalmology services described in the literature spanning from 1997 to 2020, only 6 pertained to emergency/trauma teleophthalmology care (n=6; 4.5%) and none of these specifically addressed stroke or integration into stroke care.56
Future Directions
It is our opinion that of the various technologies used to evaluate the ocular system in the emergency setting, technology enabling a fundus examination would be the most instrumental for remote triaging of acute vision loss. Non-mydriatic cameras that document the appearance of the ocular fundus are relatively easy to use and can greatly enhance the ability of practitioners to assess for non-stroke causes of acute vision loss. Several reports have published success in using nonmydriatic high-resolution digital fundus photography for obtaining fundus photos in non-ophthalmic settings such as emergency departments57,58. We believe that incorporating fundus photography with the capability of remote evaluation by ocular specialists in a telestroke consult would make a material difference in the triaging of acute vision loss.
Perhaps the most notable advantage of incorporating tele-ophthalmological evaluations into stroke-protocols for acute vision loss would be for patients presenting with acute CRAO to centers without in-person emergency ophthalmology coverage. In these cases, an urgent tele-ophthalmological evaluation and diagnosis of CRAO could help initiate transfer to a hospital with tPA capabilities where the patient may be eligible for treatment.
Artificial intelligence holds further promise for the utilization of tele-ophthalmology in a tele-stroke consult. Most tele-ophthalmology systems rely on point of care acquisition of ocular exam by non-ocular specialists and remote evaluation of the data by ocular specialists. Machine learning could aid diagnosis of ophthalmologic causes of acute vision loss even in the absence of immediate interpretation by ocular specialists. These technologies can also serve a role in diagnosis and management of patients with neurologic disease without vision loss. One study found that machine learning using retinal microvasculature features extracted from OCT-A and fundus images have the potential to diagnose acute cerebral stroke from retinal evaluation59. Using portable technologies, this could speed a diagnosis of stroke while a patient is in transit to the hospital, reducing death and disability.
Tele-ophthalmology, including evaluation of the retinal imaging, may play a role in the evaluation of and management of neurologic disease even in the absence of a diagnosis of ophthalmologic causes of vision loss. A study looking at 257 patients presenting to the emergency room with focal neurologic deficits found that the finding of arteriolar narrowing with non-mydriatic fundus imaging was an independent predictor of cerebrovascular disease overall and a diagnosis of transient ischemic attack, after controlling for the ABCD2 score and diffusion weighted imaging.58
Conclusions
In our efforts to expand patient access to specialty care through telemedicine, we must remember that our goal is to extend the reach of the medical care while ensuring noninferiority to traditional bedside treatment.60 Stroke workup is inappropriate for patients with evidence of other etiologies for their acute vision loss Working up presumed arterial occlusions without ruling out more common non-stroke causes for visual loss could lead to unnecessary testing and preclude interventions of other critical ophthalmic conditions in a timely fashion. To provide the best care for patients, common ophthalmic causes of acute vision loss should be ruled out quickly and efficiently before pursuing a stroke workup or treatment for suspected arterial occlusions. While a full ophthalmic exam is ideally performed in person at the time of the patient’s presentation for acute vision loss, tele-ophthalmology can facilitate a timely diagnosis. This is particularly important in the case of possible CRAO, where a stroke workup and even emergent thrombolytic treatment may be indicated. Few hospital centers to date offer tPA treatment to patients with CRAO. As the role of tPA treatment in patients with CRAO is better understood and may become standard of care, stroke services may depend on the integration of teleophthalmology to provide the highest quality care.
Implementing emerging telemedicine technologies to increase the communication between emergency medicine, ophthalmology, and neurology will allow practitioners to provide better care to patients with acute vision loss regardless of location.
Disclosure
The authors report no conflicts of interest in this work.
References
1. v RJ, Wilcock AD, Schwamm LH, et al. Assessment of telestroke capacity in US hospitals. JAMA Neurol. 2020;77(8):1035–1037. doi:10.1001/JAMANEUROL.2020.1274
2. Wechsler LR, Demaerschalk BM, Schwamm LH, et al. Telemedicine quality and outcomes in stroke: a scientific statement for healthcare professionals from the American heart association/American stroke association. Stroke. 2017;48(1):e3–e25. doi:10.1161/STR.0000000000000114
3. Müller-Barna P, Schwamm LH, Haberl RL. Telestroke increases use of acute stroke therapy. Curr Opin Neurol. 2012;25(1):5–10. doi:10.1097/WCO.0B013E32834D5FE4
4. Pula JH, Yuen CA. Eyes and stroke: the visual aspects of cerebrovascular disease. Stroke Vasc Neurol. 2017;2(4):210–220. doi:10.1136/SVN-2017-000079
5. Biousse V, Newman NJ. Chapter 29 Eye syndromes and the neuro‐ophthalmology of stroke. Handb Clin Neurol. 2008;93:595–611. doi:10.1016/S0072-9752(08)93029-3
6. Baltaziak K, Szpringer A, Czarnek-Chudzik A, et al. Quadrantanopia as the only symptom of post-COVID stroke in the occipital pole Case report. Medicine. 2021;100:44. doi:10.1097/MD.0000000000027542
7. Yew KS, Cheng E. Acute Stroke Diagnosis; 2009. Available from: http://www.ninds.nih.gov/doctors/NIH_Stroke_Scale.pdf.
8. Stuart-Shor EM, Wellenius GA, Dellolacono DM, Mittleman MA. Gender differences in presenting and prodromal stroke symptoms. Stroke. 2009;40(4):1121–1126. doi:10.1161/STROKEAHA.108.543371
9. Rowe FJ; VIS Group2 3. Accuracy of referrals for visual assessment in a stroke population. Eye. 2011;25(2):161. doi:10.1038/EYE.2010.173
10. Wijesundera C, Vingrys AJ, Wijeratne T, Crewther SG. Acquired visual deficits independent of lesion site in acute stroke. Front Neurol. 2020;11:705. doi:10.3389/FNEUR.2020.00705/BIBTEX
11. Rowe FJ, Wright D, Brand D, et al. A prospective profile of visual field loss following stroke: prevalence, type, rehabilitation, and outcome. Biomed Res Int. 2013;2013:1–12. doi:10.1155/2013/719096
12. Johnson LN, Baloh FG. The accuracy of confrontation visual field test in comparison with automated perimetry. J Natl Med Assoc. 1991;83(10):895.
13. Leavitt JA, Larson TA, Hodge DO, Gullerud RE. The incidence of central retinal artery occlusion in Olmsted County, Minnesota. Am J Ophthalmol. 2011;152(5):820–823.e2. doi:10.1016/J.AJO.2011.05.005
14. mac GB, Bao B, Schrag M, et al. Management of central retinal artery occlusion a scientific statement from the American heart association. Stroke. 2021;52:282–294. doi:10.1161/STR.0000000000000366
15. Hayreh SS, Podhajsky PA, Zimmerman MB. Branch retinal artery occlusion: natural history of visual outcome. Ophthalmology. 2009;116(6):1188. doi:10.1016/J.OPHTHA.2009.01.015
16. Petzold A, Islam N, Hu HH, Plant GT. Embolic and nonembolic transient monocular visual field loss: a clinicopathologic review. Surv Ophthalmol. 2013;58(1):42–62. doi:10.1016/J.SURVOPHTHAL.2012.02.002
17. Coutts SB. Diagnosis and management of transient ischemic attack. Continuum. 2017;23(1):82.
18. Lavin P, Patrylo M, Hollar M, Espaillat KB, Kirshner H, Schrag M. Stroke risk and risk factors in patients with central retinal artery occlusion. Am J Ophthalmol. 2019;200:271–272. doi:10.1016/J.AJO.2019.01.021
19. Wu CM, McLaughlin K, Lorenzetti DL, Hill MD, Manns BJ, Ghali WA. Early risk of stroke after transient ischemic attack: a systematic review and meta-analysis. Arch Intern Med. 2007;167(22):2417–2422. doi:10.1001/ARCHINTE.167.22.2417
20. Biousse V, Nahab F, Newman NJ. Management of acute retinal ischemia: follow the guidelines! Ophthalmology. 2018;125(10):1597–1607. doi:10.1016/j.ophtha.2018.03.054
21. Zhang LY, Zhang J, Kim RK, et al. Risk of acute ischemic stroke in patients with monocular vision loss of vascular etiology. J Neuroophthalmol. 2018;38(3):328–333. doi:10.1097/WNO.0000000000000613
22. Fallico M, Lotery AJ, Longo A, et al. Risk of acute stroke in patients with retinal artery occlusion: a systematic review and meta-analysis. Eye. 2019;34(4):683–689. doi:10.1038/s41433-019-0576-y
23. Sharma R, Newman N, Biousse V. Conservative treatments for acute nonarteritic central retinal artery occlusion: do they work? Taiwan J Ophthalmol. 2021;11(1):16. doi:10.4103/TJO.TJO_61_20
24. Schumacher M, Schmidt D, Jurklies B, et al. Central retinal artery occlusion: local intra-arterial fibrinolysis versus conservative treatment, a multicenter randomized trial. Ophthalmology. 2010;117(7). doi:10.1016/J.OPHTHA.2010.03.061
25. Mac Grory B, Nackenoff A, Poli S, et al. Intravenous fibrinolysis for central retinal artery occlusion: a cohort study and updated patient-level meta-analysis. Stroke. 2020;51:2018–2025. doi:10.1161/STROKEAHA.119.028743/FORMAT/EPUB
26. Bruce BB, Lamirel C, Wright DW, et al. Nonmydriatic ocular fundus photography in the emergency department supplementary material refer to web version on PubMed central for supplementary material. N Engl J Med. 2011;364(4):387–389. doi:10.1056/NEJMc1009733
27. Workforce H. National and regional projections of supply and demand for surgical specialty practitioners: 2013-2025 the national center for health workforce analysis (the National Center) informs public and private-sector decision-making on the U national and regional projections of supply and demand for surgical specialty practitioners: 2013–2025 2; 2016.
28. Sorra J, Famolaro T, Naomi Dyer Yount M, Alan Smith S, Wilson S, Helen Liu M. Hospital Survey on Patient Safety Culture: 2014 User Comparative Database Report Part II: appendix A-Overall Results by Hospital Characteristics Appendix B-Overall Results by Respondent Characteristics Part III: appendix C-Trending Results by Hospital Characteristics Appendix D-Trending Results by Respondent Characteristics; 2014. Available from: www.ahrq.gov.
29. Tauber J, Ayoub S, Shah P, et al. Assessing the demand for teleophthalmology in Florida emergency departments. Telemed E-Health. 2020;26(12):1500–1506. doi:10.1089/TMJ.2019.0260
30. Wedekind L, Sainani K, Pershing S. Supply and perceived demand for teleophthalmology in triage and consultations in California emergency departments. JAMA Ophthalmol. 2016;134(5):537–543. doi:10.1001/JAMAOPHTHALMOL.2016.0316
31. Brady CJ, Eghrari AO and Labrique AB. Smartphone-based visual acuity measurement for screening and clinical assessment. JAMA. 2015;314(24):2682–2683. doi: 10.1001/jama.2015.15855
32. Aslam TM, Parry NR, Murray IJ, et al. Development and testing of an automated computer tablet-based method for self-testing of high and low contrast near visual acuity in ophthalmic patients. Graefes Arch Clin Exp Ophthalmol. 2016;254(5):891–899. doi:10.1007/s00417-016-3293-2
33. Claessens J, van Egmond J, Wanten J, Bauer N, Nuijts R, Wisse R. The accuracy of a web-based visual acuity self-assessment tool performed independently by eye care patients at home: method comparison study. JMIR Form Res. 2023;7:.e41045. doi: 10.2196/41045
34. Labiris G, Panagiotopoulou, EK, Delibasis, K et al . Validation of a web-based distance visual acuity test. J Cataract Refract Surg. 2023;49(7): 666–671. doi: 10.1097/j.jcrs.0000000000001176
35. McAnany JJ, Smith BM, Garland A, Kagen SL. iPhone-based pupillometry: a novel approach for assessing the pupillary light reflex. Optom Vis Sci. 2018;95(10):953–958. doi: 10.1097/OPX.0000000000001289
36. Mazziotti R, Carrara F, Viglione A, et al. MEYE: web app for translational and real-time pupillometry. eNeuro. 2021;8(5):ENEURO.0122–21.2021. doi:10.1523/ENEURO.0122-21.2021.
37. Goseki T, Kunimi K, Shioya N, et al. New device for taking nine-directional ocular photographs: “9Gaze” application. JEMR. 2022;15(1):
38. Dunbar GE, Shen B, Aref A. The Sensimed Triggerfish contact lens sensor: efficacy, safety, and patient perspectives. OPTH. 2017;11:875–882. doi: 10.2147/OPTH.S109708
39. Wu Y, Luttrell I, Feng S, et al. Development and validation of a machine learning, smartphone-based tonometer. Br J Ophthalmol. 2020;104(10):1394–1398. doi: 10.1136/bjophthalmol-2019-315446
40. Russo A, Mapham W, Turano R, et al. Comparison of smartphone ophthalmoscopy with Slit-Lamp Biomicroscopy for grading vertical cup-to-disc ratio. J Glaucoma.2016;25(9):e777–e781. doi: 10.1097/IJG.0000000000000499
41. Truong P, Phan A, Truong B, Suen B, Melles G, Talke F. A smartphone attachment for remote ophthalmic slit lamp examinations. Microsyst Technol. 2020;26(11):3403–3407. doi: 10.1007/s00542-020-04894-7
42. González-Márquez F, Luque-Romero L, Ruiz-Romero MV, et al. Remote ophthalmology with a smartphone adapter handled by nurses for the diagnosis of eye posterior pole pathologies during the COVID-19 pandemic. J Telemed Telecare. 2023;29(6):474–483. doi: 10.1177/1357633X21994017
43. Russo A, Morescalchi F, Costagliola C, Delcassi L, Semeraro F. A novel device to exploit the Smartphone Camera for Fundus Photography. J Ophthalmol. 2015;2015:1–5. doi: 10.1155/2015/823139
44. Vilela MA, Valença FM, Barreto PK, Amaral CE, Pellanda LC. Agreement between retinal images obtained via smartphones and images obtained with retinal cameras or fundoscopic exams – systematic review and meta-analysis. OPTH. 2018. 12:2581–2589. doi: 10.2147/OPTH.S182022
45. Sivaraman A, Nagarajan S, Vadivel S, et al. A novel, smartphone-based, teleophthalmology-enabled, widefield fundus imaging device with an autocapture algorithm. Trans Vis Sci Tech. 2021;10(12):21. doi: 10.1167/tvst.10.12.21
46. Kim TN, Myers, F, Reber, C et al . A smartphone-based tool for rapid, portable, and Automated Wide-Field Retinal Imaging. Trans Vis Sci Tech. 2018;7(5):21. doi: 10.1167/tvst.7.5.21
47. McLaughlin D, Munshi H, Savatovsky E, Vanner E, Chang TA, Grajewski AL. Visual Field Testing in a Telehealth Setting: Remote Perimetry Using a Head-Mounted Device in Normal Eyes. Invest Ophthalmol Vis Sci. 2022;63(7):1265 – A0405.
48. Jones PR, Campbell P, Callaghan T, et al. Glaucoma home monitoring using a Tablet-Based Visual Field Test (eyecatcher): an assessment of accuracy and adherence over 6 months. Am J Ophthalmol. 2021; 223:42–52. doi: 10.1016/j.ajo.2020.08.039
49. Stapelfeldt J, Kucur ŞS, Huber N, Höhn R, Sznitman R. Virtual reality–based and conventional visual field examination comparison in healthy and glaucoma patients. Trans Vis Sci Tech. 2021;10(12):10. doi: 10.1167/tvst.10.12.10
50. Tsapakis S, Papaconstantinou D, Diagourtas A, et al . Home-based visual field test for glaucoma screening comparison with Humphrey perimeter. Clin Ophthalmol. 2018;12:2597–2606. doi: 10.2147/OPTH.S187832
51. Kong YX, He M, Crowston JG, Vingrys AJ. A comparison of perimetric results from a tablet perimeter and Humphrey field analyzer in glaucoma patients. Trans Vis Sci Tech. 2016;5(6):2. doi: 10.1167/tvst.5.6.2
52. Yang J, Liu L, Campbell JP, Huang D, Liu G. Handheld optical coherence tomography angiography. Biomed Opt Express. 2017;8(4):2287. doi: 10.1364/BOE.8.002287
53. Kim S, Crose M, Eldridge WJ, Cox B, Brown WJ, Wax A. Design and implementation of a low-cost, portable OCT system. Biomed Opt Express. 2018;9(3):1232. doi: 10.1364/BOE.9.001232
54. Sommer AC, Blumenthal EZ. Telemedicine in ophthalmology in view of the emerging COVID-19 outbreak. Graefes Arch Clin Exp Ophthalmol. 2020;258(11):2341–2352. doi:10.1007/S00417-020-04879-2
55. Meshkin RS, Armstrong GW, Hall NE, Rossin EJ, Hymowitz MB, Lorch AC. Effectiveness of a telemedicine program for triage and diagnosis of emergent ophthalmic conditions. Eye. 2022;37(2):325–331. doi:10.1038/s41433-022-01940-8
56. Walsh L, Hong SC, Chalakkal RJ, Ogbuehi KC. A systematic review of current teleophthalmology services in New Zealand compared to the four comparable countries of the United Kingdom, Australia, United States of America (USA) and Canada. Clin Ophthalmol. 2021;15:4015. doi:10.2147/OPTH.S294428
57. Pérez MA, Bruce BB, Newman NJ, Biousse V. The use of retinal photography in non-ophthalmic settings and its potential for neurology. Neurologist. 2012;18(6):350. doi:10.1097/NRL.0B013E318272F7D7
58. Vuong LN, Thulasi P, Biousse V, et al. Ocular fundus photography of patients with focal neurologic deficits in an emergency department. Neurology. 2015;85(3):256–262. doi:10.1212/WNL.0000000000001759
59. Pachade S, Coronado I, Abdelkhaleq R, et al. Detection of stroke with retinal microvascular density and self-supervised learning using OCT-A and Fundus imaging. J Clin Med. 2022;11(24):7408. doi:10.3390/JCM11247408
60. Kummervold PE, Johnsen JAK, Skrøvseth SO, Wynn R. Using noninferiority tests to evaluate telemedicine and E-health services: systematic review. J Med Internet Res. 2012;14(5):e132. doi:10.2196/JMIR.2169
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

