Back to Journals » International Medical Case Reports Journal » Volume 17
Case Reports of Severe Paediatric Sickle Cell Retinopathy: Disease Manifestations, Progression and Treatments
Received 14 June 2024
Accepted for publication 24 July 2024
Published 2 August 2024 Volume 2024:17 Pages 733—738
DOI https://doi.org/10.2147/IMCRJ.S467510
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Evdokia Sourla, Peck Lin Lip
Birmingham and Midland Eye Centre, Sandwell & West Birmingham Hospitals NHS Trust, Birmingham, UK
Correspondence: Peck Lin Lip, Email [email protected]
Abstract: Severe sickle retinopathy is commonly known in adults but not in children, hence any related treatment for sickle retinopathy in children was not well described. We reported 2 paediatric sickle patients (aged 12 and 13) presented with severe sickle retinopathy and described details of their disease progression and treatments over 2– 3 years, along with the challenges faced when managing this particular group of young age sickle cell patients. Our case reports also demonstrated the benefits of laser photocoagulation treatment to early sickle proliferative disease, and how complications from advanced severe retinopathy hindered effective treatments.
Keywords: children, seafans, vitreous hemorrhage, laser, traction, salmon patch
Introduction
Sickle cell disease (SCD) is an inherited blood disorder primarily affecting African descendants.1 The clumping of abnormal sickle-shaped hemoglobin causes vascular occlusion, reduces oxygenation and blood supply to specific body parts, leading to complications such as localized organ necrosis and commonly severe pain experienced by patients during crisis. In the case of the eyes, it may cause asymptomatic painless retinal ischemic change which could lead to retinal neovascularization (known as seafans lesion of proliferative sickle retinopathy) and vitreous hemorrhage and blindness.1,2
The prevalence of proliferative sickle retinopathy is higher in sickle patients with the HbSC genotype than the HbSS genotype.1 Published literature showed children as young as age 10 have evidence of sickle retinopathy, but severe proliferative retinopathy was reported as low as 5.6%, incidence of vitreous hemorrhage in children was even rarer.3
Although retinal laser photocoagulation was the preferred treatment for proliferative sickle retinopathy in adult sickle cell patients, the decision for treatment is challenged by the fact that seafans lesions may undergo spontaneous auto-infarction in as high as 60% of cases.4,5 Herein, paediatric patients with sickle retinopathy were often left untreated and deemed unnecessary.5 Hence information and treatment guidance on paediatric sickle retinopathy remain lacking. We reported 2 paediatric sickle patients presented with severe sickle retinopathy, described details of their disease progression and treatments over 2–3 years, along with the challenges faced when managing this particular group of young age sickle cell patients. Our report follows the Goldberg Classifications for proliferative sickle retinopathy severity (Table 1).6
 |
Table 1 Goldberg Proliferative Sickle Retinopathy Staging (PSR) to Categorize Severity of Sickle Retinopathy |
Cases Presentation
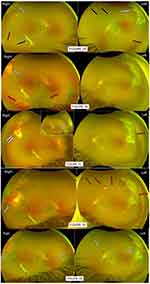
Patient 1 is known to have sickle genotype SC; at aged 13 was first seen by optician and referred with queried temporal retina lesions and hazy views in both eyes. First clinic attendance was 8 months later, confirmed good vision of Snellen 6/6 each eye but history of right eye frequent floaters. Initial examination confirmed existing advanced sickle retinopathy in peripheral retinae of both eyes, with main concern of large fibrosed seafan retinal neovascularization already on traction but flat retina (Figure 1A). There was no evidence of vitreous hemorrhage in either eye, hence no treatment was offered at this initial stage. Maculae were also normal.
Over the course of 1 year, patient experienced recurrence vitreous hemorrhage in his right eye from existing traction scars with no retinal detachment and no new active seafan, worse recorded vision over this period had been 6/60 (Figure 1B and C). Left eye vitreous hemorrhage happened later, bled from traction as well as from new active seafans (Figure 1D and E). Barrier laser photocoagulations were first performed to each tractional seafan fibrotic scars in supero-temporal peripheral, to reduce the risk of retinal detachment in future traction bleeds. At the last clinic review seven months following the initial laser treatment, although old vitreous hemorrhage still to resolve, but patient reported no further re-bleeds in his right eye, vision recovered well and maintained at Snellen 6/7 in each eye. Additional laser treatment was repetitively performed at each clinic visit to any observed new seafans or residual activeness of treated seafans.
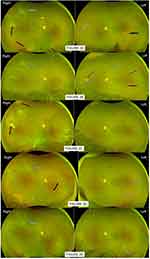
Patient 2 has sickle genotype SC, at aged 12 and asymptomatic, optician referred with queried right eye retinal detachment and left eye retinal lesions. Initial assessment confirmed excellent Snellen vision at 6/5 despite evidence of advanced fibrosed seafan neovascularization on traction and significant vitreous hemorrhage in his Right eye. His left eye had less severe tractional seafan neovascularization, a salmon patch but no vitreous hemorrhage (Figure 2A). Maculae were normal. With some obscured view from vitreous bleed, limited barrier laser treatment was nevertheless performed on the tractional seafans in Right eye.
The salmon patch in left eye resolved in 4 months, with more new salmon patches developed in different parts of the retina of the same eye (Figure 2B). Over the period of 2.5 years, patient 2 frequently missed scheduled clinic visits (attendance rate = 45%). There were recurrent vitreous hemorrhage in Right eye with worst recorded visual level of counting fingers from his right eye when he eventually re-attended eye casualty. However, patient’s right eye vision did improve to normal at 6/6 at most clinic visits despite dense vitreous hemorrhage which was slow to clear obscuring most of peripheral retina view (Figure 2C and D). Additional laser to Right eye was hence not possible due to non-clearing peripheral hemorrhage. However, barrier laser/sector laser were performed more easily to Left eye when new sprouting seafans were found at each clinic visit, before any vitreous hemorrhage happened (Figure 2D). Keeping good vision of 6/7 right, 6/5 left, lacking window opportunity for effective laser treatment through the unclearing peripheral vitreous hemorrhage, patient eventually developed a localized peripheral tractional retinal detachment in his right eye, which had enough surrounding barrier laser to be secured and non-progressive at the last clinic visit (Figure 2E).
Discussion
Previous published literature confirmed that children with SCD could develop proliferative sickle retinopathy but severe blinding disease is rarely reported in children with sickle cell.5,7 Our case reports confirmed that sickle cell children as young as age 12 could have severe advanced sickle retinopathy. Indeed, the severe retinopathy was already manifested in the first clinic examination, indicating that retinopathy development must have started years before. Visual level is a poor indication to reflect severity of sickle retinopathy. Majority of sickle cell patients (both adults and children) have good vision with no symptoms until retinopathy is too advanced causing symptoms of floaters or visual loss due to vitreous hemorrhage as in our case reports. This is due to sickle retinopathy disease clinical signs and features manifest predominantly in the far peripheral retina, hence often undetected by standard imagings used by optician. With the availability of advanced fundus imagings in recent years, in particular, the non-invasive wide-field fundus photography provides easy and clearer detection of peripheral sickle retinopathy disease.8 We therefore speculate the prevalence of severe sickle retinopathy in children could be much higher than previously reported.7,9
Decision to treat sickle retinopathy is perplexing for clinicians due to lack of published literature and lack of clinical intervention treatment trials. Most published studies on treatments were based on sickle adult patients but not on sickle paediatric cohort. Although laser photocoagulation was regarded as the preferred choice to treat sickle retinopathy, guidance is unclear on applying the many different types/patterns of laser photocoagulation described.5 Although seafans could undergo auto-infarction and become inactive, their localized fibrosis often lead to tractional scars; traction bleeds contribute towards recurrent vitreous hemorrhage and the risk of retinal detachment. We described effective laser treatment to paediatric sickle retinopathy as demonstrated in our patients: “sectorial laser” is best applied when active seafan lesion is still small and not yet elevated, sector laser application is a ring of confluent laser around a seafan lesion (similar to delivering an effective “retinopexy” laser treatment for a retinal hole) (Figure 2E); “barrier laser” is best applied at the border of an elevated fibrosed seafan scar on traction (Figure 1E), to hope to reduce risk of retinal detachment with future traction bleeds.
Decision on optimal effective treatment in young patients remain a challenge due to few factors. Delivering effective laser required good cooperation of patients in which case, both our 2 school boys coped very well with the out-patient slit-lamp lasering procedures at their young age of 13. Understandably, attending scheduled clinic visits would be unlikely priorities in their busy schooling activities, when they also benefited more time of good vision than period affected by bad vision as vitreous hemorrhage seem to clear faster in center than in peripheral. In addition, good vision and painless nature of sickle retinopathy often render patients unaware of seriousness of the eye disease, subsequently denying the opportunity of optimal treatment window and eventuality of disease progression. We nevertheless had clinic reviews arranged during school holiday weeks to encourage needed attendances.
The other challenging factor is getting children to recognize their visual symptoms. Children are less likely to report to their parents or teachers unless the disease causes symptoms in both eyes; unilateral visual changes is often ignored. Herein, retinopathy screening at an early age could help eliminate late assessment from delayed reporting of symptoms. Published literature had suggested retinopathy screening to start as young as age 9 in children with SCD.3,9 Our case reports would support this concept as our 2 patients had already presented with advanced tractional scars at age 12 and 13, hence speculating proliferative sickle retinopathy development would indeed have started at a much earlier age.
Conclusion
In summary, children as young as age 12 can progress to develop advance sickle retinopathy if untreated. As in the adults cohort, severe sickle retinopathy in children is preventable if there is an early window of recognition and optimal treatment opportunity. Our case reports detailed management and laser treatment for paediatric sickle retinopathy which is very much lacking in available literature. Optimal management plan for this young age group remains a challenge, taking into consideration of schooling timing and cooperation of individual child for laser treatment. Our case-reports nevertheless provide some evidence of the need to start sickle retinopathy screening programme at earlier school age, perhaps inco-operating into the existing visual screening programme for childhood amblyopia/childhood squints (at around school age of 5 years old). With the easily available, non-invasive advanced investigational tool such as ultra-widefield fundus photography, sickle retinopathy screening programme at younger age could indeed be very feasible and effective. This prospect also leads us to the much needed research and studies that can better understand sickle retinopathy treatment choices and effectiveness in both adults and paediatrics.
Ethics Approval and Consents for Publication
This ethical approval for this retrospective case series report was granted from the Institutional Review Board (Sandwell and West Birmingham Research and Development review board) in accordance with the “Good Clinical Practice” regulations in the United Kingdom and adhered to the tenets of the Declaration of Helsinki. Informed written parental consents were obtained from all paediatric patients for investigations/treatment procedures as part of the routine and standard clinical care in our real-world clinical practice. Relevant fundus photography and angiographic images were anonymized with patients’ consents/approvals to publish.
We also obtained from parents of our two paediatric patients on specific consents/approvals for this article and relevant fundus photography images to be used for publication.
Disclosure
The authors received no funding for this work, and declared no conflicts of interest related to this work.
References
1. Van Meurs JC. Relationship between peripheral vascular closure and proliferative retinopathy in sickle cell disease. Graefes Arch Clin Exp Ophthalmol. 1991;229(6):543–548. doi:10.1007/BF00203319
2. Downes SM, Hambleton IR, Chuang EL, et al. Incidence and natural history of proliferative sickle cell retinopathy: observations from a cohort study. Ophthalmology. 2005;112:1869–1875. doi:10.1016/j.ophtha.2005.05.026
3. Babalola OE, Wambebe CO. When should children and young adults with sickle cell disease be referred for eye assessment? Afr J Med Med Sci. 2001;30(4):261–263.
4. Penman AD, Serjeant GR. Recent advances in the treatment of proliferative sickle cell retinopathy. Curr Opin Ophthalmol. 1992;3(3):379–388. doi:10.1097/00055735-199206000-00013
5. Myint KT, Sahoo S, Thein AW, Moe S, Ni H. Laser therapy for retinopathy in sickle cell disease. Cochrane Database Syst Rev. 2015;2015(10):CD010790. doi:10.1002/14651858.CD010790.pub2
6. Goldberg MF. Classification and pathogenesis of proliferative sickle cell retinopathy. Am J Ophthalmol. 1971;71(3):649–665. doi:10.1016/0002-9394(71)90429-6
7. Children With Sickle Cell Disease Appear to Suffer Eye Complications at Same Rate as Adults, American Academy of Ophthalmology, Available from: https://www.aao.org/newsroom/news-releases/detail/children-with-sickle-cell-disease-appear-to-suffer.
8. Pahl DA, Green NS, Bhatia M, Chen RWS. New ways to detect pediatric sickle cell retinopathy: a comprehensive review. J Pediatr Hematol Oncol. 2017;39(8):618–625. doi:10.1097/MPH.0000000000000919
9. Li J, Bender L, Shaffer J, Cohen D, Ying GS, Binenbaum G. Prevalence and onset of pediatric sickle cell retinopathy. Ophthalmology. 2019;126(7):1000–1006. doi:10.1016/j.ophtha.2019.02.023
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.