Back to Journals » Patient Preference and Adherence » Volume 18
“I Was Almost in Disbelief” - Qualitative Analysis of Experiences and Expectations Among Psoriasis Patients Undergoing Biologic Treatment with Secukinumab
Authors Hölsken S , Benthin C, Krefting F, Mühlhaus S, Nestoriuc Y, Schedlowski M, Sondermann W
Received 19 February 2024
Accepted for publication 29 May 2024
Published 24 June 2024 Volume 2024:18 Pages 1299—1310
DOI https://doi.org/10.2147/PPA.S458643
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Johnny Chen
Stefanie Hölsken,1,* Carina Benthin,2,3,* Frederik Krefting,4 Senta Mühlhaus,1 Yvonne Nestoriuc,2,3 Manfred Schedlowski,1,5 Wiebke Sondermann4
1Institute of Medical Psychology and Behavioral Immunobiology, University Hospital Essen, University of Duisburg-Essen, Essen, Germany; 2Clinical Psychology, Helmut-Schmidt-University/University of the Federal Armed Forces Hamburg, Hamburg, Germany; 3Systems Neuroscience, University Medical Center Hamburg-Eppendorf, Hamburg, Germany; 4Department of Dermatology, Venereology and Allergology, University Hospital Essen, University of Duisburg-Essen, Essen, Germany; 5Department of Clinical Neuroscience, Osher Center for Integrative Medicine, Karolinska Institutet, Stockholm, Sweden
*These authors contributed equally to this work
Correspondence: Wiebke Sondermann, Department of Dermatology, Venereology and Allergology, University Hospital Essen, University Duisburg-Essen, Hufelandstr. 55, Essen, 45122, Germany, Tel +49201-723-3647, Fax +49201-723-5409, Email [email protected]
Purpose: Psoriasis is a highly debilitating chronic inflammatory disease. Increased understanding of its pathophysiology has enabled development of targeted treatments such as biologics. Several medical treatments have been shown to be influenced by patients’ experiences and expectations. However, only little is known about patients’ experiences with and expectations towards biologics. Our objectives were to identify patients’ treatment experiences and treatment expectations and assess their trajectories over the course of treatment with the IL-17A inhibitor secukinumab. Moreover, we aimed to document effects of psoriasis, factors that influence symptomatology, and prior treatment experiences.
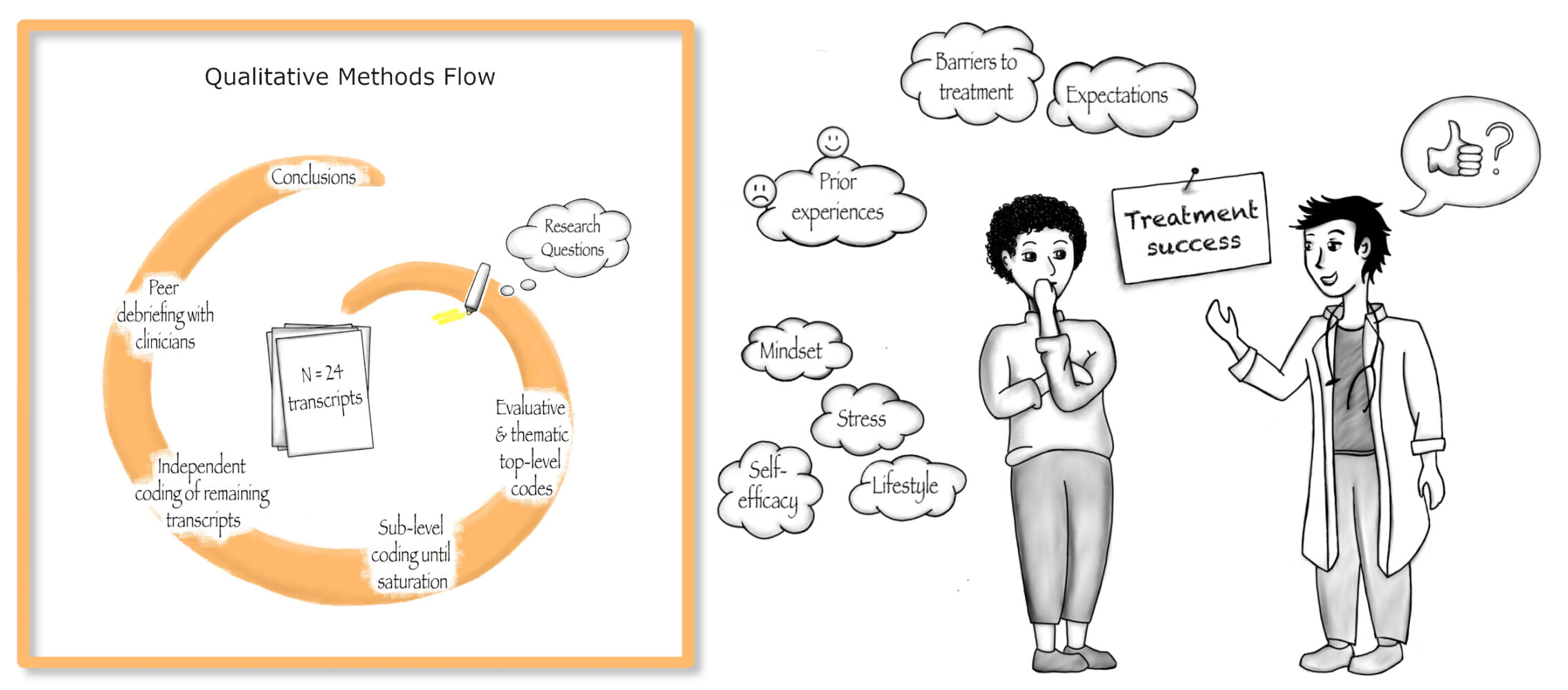
Patients and Methods: We conducted semi-structured interviews with n = 24 patients with moderate-to-severe plaque psoriasis and employed a qualitative content analysis to derive thematic and evaluative codes. Findings were validated via peer debriefings with experienced dermatologists.
Results: Patients reported burdensome physical and psychological psoriasis symptoms and identified factors that can improve or worsen symptomatology, including stress and self-efficacy. Prior treatment experiences were mostly negative. Past barriers to effective treatment included time constraints or limited access. Concerning secukinumab, patients initially expected complete to partial remission of symptoms and occurrence or absence of side effects. Closer inspection of expectations and experiences revealed three trajectories. For most patients, initial expectations were met and future expectations remained unchanged. For the other patients, however, the experience did not match their initial expectation. One group then adapted their future expectations according to their experience, while the other group did not.
Conclusion: To our knowledge, this is the first qualitative study to assess expectations towards treatment effectiveness and side effects, their trajectories, and interplay with experiences among psoriasis patients. Our findings highlight the value of further research on the subject in order to optimize care for psoriasis patients and to learn more about the trajectories and influence of treatment expectations in general.
Plain Language Summary: Patients’ expectations towards a certain treatment can influence how well it will work and whether side effects occur or not. Such effects have been shown for a wide range of conditions, including dermatological ones. For patients suffering from psoriasis, a chronic inflammatory skin disease, modern biologics offer promising treatment options. Therefore, modern biologics are likely associated with high expectations by patients. Yet, the roles of treatment expectations and the experiences of patients undergoing biologic treatment have only received little attention to date.
With this qualitative study, we aimed to understand patients’ perspectives on these treatments, the symptoms they suffer from, expectations towards treatment effectiveness and side effects as well as treatment experiences in the past and over the course of treatment with a modern biologic. To this end, we conducted and transcribed interviews with 24 patients after several months of biologic treatment. We then analyzed interview transcripts to determine underlying categories and summarized these in a common framework.
We found that patients’ debilitating physical and psychological symptoms were often not well controlled in past treatments. Still, patients had positive expectations with regard to biologic treatment. For a large group of patients, their actual experiences met their expectations, leaving them with unchanged expectations for the future. Another important finding was the good understanding of factors influencing their symptomatology that patients had, like stress and self-efficacy. The reported findings may aid doctors in their clinical work, to further improve care for psoriasis patients.
Keywords: psychodermatology, patient perspective, treatment success, doctor-patient relationship, secukinumab
Graphical Abstract:
Introduction
Psoriasis is a chronic inflammatory disease with an estimated prevalence of 2–3% in Western Europe.1–4 Patients regularly experience stigmatization and decreased quality of life.5 Psoriasis is often accompanied by comorbid somatic or psychological conditions,6 such as cardiovascular disease,7 depression, or anxiety.8,9 Stress, lifestyle choices, and environmental factors can amplify psoriasis symptomatology,10–12 contributing to a vicious cycle of symptom deterioration and increased patient burden.
Psoriasis treatments include topical therapies in patients with mild symptoms and phototherapy as well as systemic therapies in patients with moderate to severe symptomatology.13 Conventional anti-psoriatic systemic therapies have been available for a long time, but their use is impeded by a relatively limited efficacy combined with a sub-optimal side-effect profile.14–16 Increased understanding of psoriasis pathophysiology has enabled the development of biologics targeting tumor necrosis factor α (TNFα; adalimumab, infliximab, certolizumab, etanercept), interleukin (IL)-17 (secukinumab, ixekizumab, brodalumab, bimekizumab), IL-12/-23 (ustekinumab) or IL-23 (guselkumab, tildrakizumab, risankizumab) with improved effectiveness and tolerability.17–19 The monoclonal antibody secukinumab, for instance, targets Interleukin-17A and is associated with excellent skin responses20,21 and an improved side-effect profile compared to conventional systemic treatments.22 Yet, biologics are cost-intensive and, therefore, may usually only be prescribed if therapeutic alternatives did not show sufficient efficacy or cannot be used due to intolerable side effects.13
The high effectiveness of modern biologics such as secukinumab is likely to result in increased treatment expectations among patients. Expectations are an important contributor to the success of a range of medical treatments.23 On the one hand, patients’ positive treatment expectations can drive placebo effects, ie, positive treatment effects beyond the pharmacological action of an administered drug.24 The potential of patients’ expectations to enhance treatment effectiveness via the placebo effect has also been established in the field of dermatology.25 On the other hand, patients’ negative expectations can trigger nocebo effects, ie, adverse effects extending beyond the pharmacological action of a drug.26–28 Despite the potential relevance of expectations in safe and effective treatment for psoriasis, little is known about patients’ expectations towards the effectiveness and side effects of modern biologics such as secukinumab.
In recent years, patients’ views have increasingly moved into the focus of research, reflecting the adoption of a more cooperative model of doctor-patient interaction in clinical practice.29 Analyzing patients’ views can provide new insights into experiences with modern biologics and expectations towards effectiveness and side effects, allow an enhanced understanding of psoriasis in patients’ lives, and yield starting points for optimized, individualized treatment regimens. With this qualitative study, we aimed to understand how psoriasis affects patients’ lives and shed light on patients’ experiences with and expectations towards secukinumab treatment.
Materials and Methods
We conducted semi-structured interviews with patients with moderate-to-severe plaque psoriasis to understand the effects of psoriasis and identify factors that can influence symptomatology. We aimed to document patients’ prior treatment experiences and barriers to treatment. Moreover, we asked patients about their expectations towards effectiveness and side effects of secukinumab therapy and their experienced effectiveness and side effects over the course of treatment. Ultimately, we aimed to assess the interplay of patients’ experiences and expectations.
This qualitative study was conducted at the end of a 16-week randomized controlled trial (RCT) investigating how psychological factors affect response to secukinumab therapy in psoriasis patients (German Clinical Trials Register-ID: DRKS00022104).30 Within the scope of the RCT, some patients received a 75% dose reduction of secukinumab. Study conception and conduct followed Consolidated Criteria for Qualitative Research (COREQ).31 Reporting is in line with Standards for Reporting Qualitative Research (SRQR).32
Patients and Recruitment
We conducted semi-structured interviews with N = 24 patients with moderate-to-severe plaque psoriasis, who were eligible for systemic treatment with secukinumab based on the German S3-Guideline on the treatment of Psoriasis vulgaris for the initiation of biologic treatment13 and adhering to the Summary of Product Characteristics for secukinumab33 (for detailed eligibility criteria see30). Patients were recruited from the RCT and purposefully sampled to include equal proportions of participants of all experimental groups, male and female gender, age groups 18–35 years versus >35 years, and with or without prior experiences with systemic treatment. Interviews were conducted until saturation was reached, ie, no new relevant knowledge was obtained.34 Patients were approached during study visits and received standardized study information. If patients agreed to participate and provided informed consent, qualitative interviews were arranged.
Data Generation
Prior to commencement of the RCT, no relationship was established between patients and study staff. Interviews were conducted in German using a pilot-tested, semi-structured interview guide developed by SH (MSc, psychologist, PhD student) and CB (MSc, psychologist, PhD student). The interview guide can be found in Supplementary Table 1. SH arranged interviews with patients and remained their constant contact person. SH and SM (MD student) conducted and audio-recorded interviews on-site in the outpatient department of the Clinic for Dermatology, Venereology and Allergology at the University Hospital Essen, Germany. SH transcribed interviews and took field notes. To place qualitative findings in context, demographic and medical characteristics were collected.
Data Analysis
To aid our analysis, we used the qualitative research software MaxQDA.35 Analysis was conducted in German. Findings were translated into English. The initial coding frame comprised both thematic and evaluative codes directly referring to our research questions. First, CB and SH re-read all 24 transcripts and assigned top-level codes in several cycles. Second, CB and SH drafted focused case summaries for all interviews in order to develop sub-level codes.36 Third, CB and SH assigned sub-level codes in consultation with MS, WS, and YN until no new sub-level codes emerged. Fourth, CB and SH independently coded the remaining 15 transcripts, and assessed intercoder agreement using Cohen’s kappa coefficient (κ= 0.86, ie, high agreement). Ultimately, SH and CB discussed diverging codings and reached full consensus. Code definitions were discussed during peer debriefing sessions with a group of dermatologists experienced in psoriasis treatment and were refined throughout analysis.
Results
Among the 24 patients, 10 were female (41.67%). Age ranged from 22 to 73 years (M = 45.17 years, SD = 16.17 years). Duration of illness was 16.34 years on average (range 0.5 to 54 years). Patients had on average undergone 14.17 years of education (including academic and professional training, range 0 to 22 years). Within the scope of the RCT,30 seven patients received treatment as usual with secukinumab, while seventeen patients received a dose of only 25%. Sixteen patients had undergone prior systemic treatment. Four of them had previously been on treatment with a biologic (guselkumab, adalimumab, ixekizumab and etanercept, respectively); the other patients had received fumaric acid esters, methotrexate, apremilast or a systemic corticosteroid. The initial Psoriasis Area and Severity Index (PASI) among patients ranged from 4 to 28.2 (M = 16.41, SD = 5.95). Post-treatment PASI ranged from 0 to 16 (M = 4.38, SD = 3.64). PASI-change ranged from 0 to 100% (median 76.72%). Further descriptives can be found in Supplementary Table 2. In the following paragraphs, qualitative findings are documented for each top-level code, with according sub-level codes in italics. Concept-driven thematic and evaluative top-level codes are listed in Table 1. Illustrative quotations are listed in tables at the end of each section (Tables 2–7).
 |
Table 1 Overview of Concept-Driven Thematic and Evaluative Top-Level Codes |
 |
Table 2 Effects of Psoriasis: Subcodes and Exemplary Quotations |
 |
Table 3 Factors Influencing Symptomatology: Subcodes and Exemplary Quotations |
 |
Table 4 Prior Treatment Experiences: Subcodes and Exemplary Quotations |
 |
Table 5 Barriers to Treatment: Subcodes and Exemplary Quotations |
 |
Table 6 Treatment Effectiveness: Evaluative Categories, Subcodes and Exemplary Quotations |
 |
Table 7 Side Effects: Evaluative Categories, Subcodes and Exemplary Quotations |
Effects of Psoriasis
Patients experienced debilitating physical symptoms of psoriasis, including pain, itch, and skin plaques. Patients reported negative affect resulting from their disease. They experienced hopelessness, anger, low mood, and illustrated various fears relating to psoriasis. Some patients described that they felt their psoriasis was a burden to their social environment.
Patients experienced a decreased quality of life due to suffering from psoriasis. Among others, this decrease resulted from physical and emotional symptoms, restrictions in sports and activities, limited choice of clothing, and social isolation. Patients felt ashamed of the visible signs of their psoriasis. For some, plaques and flaking resulted in severe discomfort. Some patients experienced stigmatization.
Factors Influencing Symptomatology
Patients identified stress as a relevant factor influencing their physical symptoms. Increased stress resulted in symptom deterioration. Reduced stress and active relaxation strategies were associated with symptom improvement. Patients described that their mental wellbeing also influenced their physical symptoms, and wished that treatment plans considered this relationship, for example, within integrated treatment approaches or through counselling and psychotherapeutic support.
Some patients illustrated that a high sense of self-efficacy, that is, knowing strategies to improve their health state and feeling capable of applying these, enabled counteracting symptom deterioration. Patients perceived that disease acceptance mitigated effects of psoriasis. A healthy lifestyle positively affected physical symptoms and negative affect. Some patients described an upward spiral concerning effective symptom reduction: If a certain treatment was effective, patients’ quality of life and sense of self-efficacy increased and enabled them to apply better and regular self-care strategies. In turn, this further enhanced positive treatment effects and symptom reduction.
External factors influencing patients’ symptomatology included a positive treatment setting, social support, and environmental factors. Patients experienced that a positive treatment setting positively affected their course of treatment. A positive treatment setting was characterized by a positive doctor-patient-relationship including a warm and competent practitioner, no time pressure, and feeling taken seriously. Ultimately, patients perceived that social support within their close environment and work context and environmental factors such as the season and weather could exert a positive influence on their symptomatology.
Prior Treatment Experiences
Patients, who had tried different treatments, often experienced a lack of treatment success. Treatments such as creams, light therapy, or bathing were either not effective or only lead to insignificant improvement of symptoms. Yet, some patients continued such treatments for several years. Some treatments were effective, even resulting in total clearance of the skin, but only yielded short-term treatment success. These treatments either stopped working, were discontinued by patients due to the associated time and effort, or had to be discontinued due to complications or side effects. Patients had experienced various adverse events and complications. Systemic treatments were associated with debilitating side effects such as severe gastrointestinal symptoms. Use of topicals was accompanied by complications including discomfort due to texture, limited choice of clothing, or difficulties when treating the scalp.
Patients illustrated heterogeneous experiences regarding their relationships with healthcare professionals. Positive experiences included competent and empathic treatment or referral to the right specialists. Negative experiences included a perceived lack of competence and empathy. Some patients reported that they had received an incorrect diagnosis and, consequently, adhered to unnecessary, ineffective treatments for years. Some patients stated that healthcare professionals had emphasized not having much experience with psoriasis treatment and specific medications and therefore refused a desired treatment.
Some patients resorted to self-treatment strategies and used natural or home-made care products, established rituals such as dedicated wellbeing-days, or changed their diet to counteract psoriasis symptoms. Multiple patients referred to treatment experiences of others within their social environment or on social media as having affected their own prior treatment choices.
Barriers to Treatment
Patients reported several hindrances to effective treatment options. Central problems were time and effort related to a certain treatment. Some treatments were perceived as too time-consuming or difficult to integrate with work and social life. Furthermore, some patients had no or difficult access to a desired treatment. Reasons for this included a specific treatment not being available in the region, treatment not being prescribed or only being prescribed in insufficient amounts, or long waiting lists in specialized centers. Some patients reported non-utilization of an offered treatment. Ultimately, in some patients, treatment was hindered by the presence of comorbidities.
Interplay of Expectations and Experiences Concerning Perceived Treatment Effectiveness and Side Effects
At study start, patients either expected complete remission (n = 7) or partial remission of symptoms (n = 14). Over the course of treatment, some patients reported having experienced profound treatment success (n = 11), others moderate treatment success (n = 10), and one patient no treatment success at all. Later expectations towards further effectiveness included profound treatment success (n = 9), moderate treatment success (n = 10), or no treatment success at all (n = 1). Some patients made no or ambiguous statements, which were coded as non-quantifiable and excluded from further analysis.
Patients could generally be grouped into three clusters. The first group comprised patients whose experienced effectiveness matched their expectations at the start of the study. Over the course of treatment, their expectations remained unchanged (n = 8). The second cluster encompassed patients whose experienced effectiveness did not match their initial expectation. Over the course of treatment, their expectation was adapted according to their experience (n = 7). The third group included patients whose experienced effectiveness did not match their initial expectations, but who did not adjust their expectations accordingly (n = 5).
At study start, patients either expected that side effects would occur (n = 14) or would not occur (n = 9). Over the course of treatment, some patients experienced side effects (n = 4), while most did not experience any (n = 19). Later expectations towards further side effects included occurrence of side effects (n = 4) or absence of side effects (n = 16). Some patients made no or ambiguous statements, which were coded as non-quantifiable and excluded from further analysis.
Again, patients could be grouped into three clusters. The first group included patients whose experience matched their initial expectation. Over the course of treatment, their expectation concerning the occurrence of side effects remained unchanged (n = 9). The second cluster comprised patients whose experience concerning the occurrence of side effects did not match their expectation at study start, as they did not experience any. Over the course of treatment, their expectation was adapted according to their experience (n = 8). The third group included patients whose experience concerning side effects did not match their initial expectations, but who did not adjust their expectations (n = 4).
Discussion
The aim of this qualitative study was to understand how psoriasis affects patients’ lives and to characterize patients’ treatment experiences and expectations towards secukinumab treatment.
Our findings underline the large, negative impact of psoriasis on patients’ lives reported in existing literature and largely replicate factors influencing symptomatology.37 Among our sample, patients also felt weighed down by their mental load, experienced stigmatization, and resented their own appearance. Several of these consequences of psoriasis are addressed in the commonly employed Dermatology Life Quality Index (DLQI38), underscoring the tool’s relevance. At the same time, patients’ knowledge of factors influencing symptomatology indicated a good understanding of their disease and resourcefulness in dealing with psoriasis. Resources like self-efficacy and self-management skills can exert a positive influence on health-related quality of life,39,40 potentially counteracting the negative impact of psoriasis. However, patients’ resourcefulness partly resulted from feeling left alone or inadequately treated by healthcare providers.
Patients had faced barriers that withheld them from receiving appropriate care in the past. In line with Eissing et al,41 we identified barriers related to availability of treatments, fear of side effects and/or inconveniences, time constraints, and comorbidities. Inadequate treatment and insufficient quality of care remain a key obstacle to reducing the impact of psoriasis on patients’ lives.42
Expectations towards effectiveness of secukinumab treatment were moderate to high. In some cases, prior experiences reported by others had influenced treatment expectations. More than half of all patients expected to experience side effects throughout the trial. This expectation was partly based on prior experiences of side effects caused by other treatments.
In terms of the placebo effect,27 positive treatment expectations regarding modern biologics may generally be regarded as beneficial. Nevertheless, overly positive treatment expectations may be problematic. Psoriasis patients have often experienced disappointment over unmet expectations during previous treatments.37 Within our sample, one participant had expected treatment success but did not perceive any effectiveness. While the resulting negative adjustment of expectations in this case may have been realistic, it could present a barrier to appropriate care and treatment adherence in the future. Concerning side effects, Eissing et al concluded that it is particularly important to inform patients with psoriasis thoroughly and realistically about occurrence and severity.41 To avoid nocebo effects, however, it is important to keep expectations about side effects as low as possible43 and provide additional psychoeducation.44 Considering the therapeutic algorithms as defined in the guidelines,13,45 patients who receive modern biologics typically have undergone less effective and less tolerable prior treatments. Negative prior experiences can reinforce negative expectations, thereby enhancing the nocebo effect.27 Our results indicate the need to further investigate expectations among psoriasis patients and examine the potential for targeted reinforcement of positive and modification of negative expectations.
Regarding trajectories of experiences and expectations, several patients did not adjust their expectations according to their experience. Patients who did not show a positive adjustment of expectations despite a positive treatment experience may benefit less from treatment in the long term. Patients who reported a positive adjustment of expectations despite moderate treatment effectiveness may have greater benefit from treatment. Regarding possible predictors of inter-individual differences in the adjustment of expectations, cross-tabulation was not informative. Future studies should investigate possible predictors and consequences of non-adjustment of expectations.
To our knowledge, this is the first qualitative study to assess expectations concerning treatment effectiveness and side effects, their trajectories, and interplay with experiences among psoriasis patients. With in-depth interviews from 24 patients recruited using a purposive sampling strategy for maximum sample variation, our findings stem from a sound qualitative data basis.
The potential of utilizing patients’ treatment expectations to enhance positive and prevent negative treatment effects is apparent in the field of dermatology.25 Our findings contribute to the emerging literature on the effects of psoriasis on patients’ lives and the factors affecting symptomatology and adds to the barriers to appropriate care described so far.41,46 Peer debriefing further enhanced clinical relevance of our findings.
However, the following limitations should be noted: Expectations had to be assessed retrospectively due to otherwise possible confounding of the RCT. An additional question on patients’ self-assessed quality of memory could have helped to boost reliability. Nonetheless, prospective and retrospective assessments could be shown to possess moderate to high concordance in the case of other constructs.47 Based on our results, future studies employing a prospective assessment in addition to a retrospective one seem promising to compare expectations before and after treatment. In addition, such a design could test in how far interviewing patients about their expectations prospectively versus retrospectively may affect expectations differently. Sampling may have favored patients with more positive treatment expectations, as patients participating in the RCT were highly motivated and started treatment with moderate to high expectations concerning effectiveness. However, as all approached patients consented to giving the interview, no additional bias was introduced. As patients in this study only received treatment with secukinumab, the question in how far findings apply to treatment with other biologics needs to be elucidated in future projects. Expectations concerning side effects were assessed binarily as either present or not. A more detailed, prospective assessment of expectations as well as the triangulation of qualitative findings with appropriate quantitative measures may be a promising direction for future studies.
Conclusion
This study characterized expectations towards as well as their trajectories over the course of secukinumab treatment in psoriasis patients. Expectations towards effectiveness of secukinumab treatment were moderate to high and more than half of all patients expected to experience side effects. Despite deviating experiences, several patients did not adjust their expectations over the course of treatment. Patients who do not show a positive adjustment of expectations despite a positive treatment experience may benefit less from treatment in the long term, while patients who report a positive adjustment of expectations despite moderate treatment effectiveness may have greater benefit from treatment. These observations may serve as an important starting point for future research into the interplay of experiences and expectations.
Our findings highlight some important points for the clinical practice: In light of negative experiences, patients wished to be treated by their healthcare professionals in a competent and empathic manner. Tailored communication training could aid formation of a trusting patient-provider relationship. Effects of psoriasis extending beyond the skin, such as stigmatization and psychological comorbidities, should be routinely assessed and patients’ mental well-being should be considered using both standardized tools such as the DLQI and empathic communication. Factors influencing psoriasis symptomatology and barriers to treatment should be discussed to increase patients’ self-efficacy and foster adherence. Patients also reported the wish for an integrated treatment approach that incorporates psychosocial aspects of suffering from psoriasis.
Taken together, our results underscore the value of further research on the topic in order to optimize the overall care for psoriasis patients. Better understanding of patients’ expectations as well as their changes over the course of therapy can foster communication between patients and physicians and thereby support adherence.
Abbreviations
IL, Interleukin; TNF, tumor necrosis factor; PASI, Psoriasis Area and Severity Index; RCT, randomized controlled trial; DLQI, Dermatology Life Quality Index.
Data Sharing Statement
The data that support the findings of this study are available from the authors upon reasonable request.
Ethics Approval and Informed Consent
The study was approved by the institutional ethics committee of the University Duisburg-Essen (19-8636-BO). It was conducted in accordance with the Declaration of Helsinki. All patients gave their written informed consent to study participation, including their consent to the publication of anonymized responses.
Acknowledgments
We thank all patients who participated in our interviews and shared their valuable experiences. We thank Dr Katharina Baratella for supporting us with her expert knowledge on psoriasis.
Funding
Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – Project-ID 422744262 – TRR 289. Gefördert durch die Deutsche Forschungsgemeinschaft (DFG) – Projektnummer 422744262 – TRR 289. The funder was not involved in planning or conducting of this research project.
Disclosure
Frederik Krefting has received travel support and/or consulting fees from Almirall, Boehringer Ingelheim and Novartis outside the submitted work. Wiebke Sondermann reports grants and/or travel support and/or personal fees and/or speaker honoraria from medi GmbH Bayreuth, Abbvie, Almirall, Amgen, Bristol-Myers Squibb, Celgene, GSK, Janssen, LEO Pharma, Lilly, MSD, Novartis, Pfizer, Roche, Sanofi Genzyme and UCB outside the submitted work. Manfred Schedlowski reports speaker honoraria from Almirall, Lilly, Bristol-Myers Squibb, Janssen, Berlin Chemie, Abbvie, and Mylan outside of the present publication. The other authors have no conflicts of interest to declare for this work.
References
1. Schafer I, Rustenbach SJ, Radtke M, Augustin J, Glaeske G, Augustin M. Epidemiologie der Psoriasis in Deutschland – Auswertung von Sekundärdaten einer gesetzlichen Krankenversicherung[Epidemiology of psoriasis in Germany--analysis of secondary health insurance data]. Gesundheitswesen. 2011;73(5):308–313. German. doi:10.1055/s-0030-1252022
2. Parisi R, Iskandar IYK, Kontopantelis E, Augustin M, Griffiths CEM, Ashcroft DM. National, regional, and worldwide epidemiology of psoriasis: systematic analysis and modelling study. BMJ. 2020;369:m1590. doi:10.1136/bmj.m1590
3. Griffiths CEM, Armstrong AW, Gudjonsson JE, Barker J. Psoriasis. Lancet. 2021;397(10281):1301–1315. doi:10.1016/S0140-6736(20)32549-6
4. Patruno C, Napolitano M, Balato N, et al. Psoriasis and skin pain: instrumental and biological evaluations. Acta Derm Venereol. 2015;95(4):432–438. doi:10.2340/00015555-1965
5. Finlay AY, Kelly SE. Psoriasis--an index of disability. Clin Exp Dermatol. 1987;12(1):8–11. doi:10.1111/j.1365-2230.1987.tb01844.x
6. Nestle FO, Kaplan DH, Barker J. Psoriasis. N Engl J Med. 2009;361(5):496–509. doi:10.1056/NEJMra0804595
7. Boehncke WH. Systemic inflammation and cardiovascular comorbidity in psoriasis patients: causes and consequences. Front Immunol. 2018;9:579. doi:10.3389/fimmu.2018.00579
8. Hölsken S, Krefting F, Schedlowski M, Sondermann W. Common fundamentals of psoriasis and depression. Acta Derm Venereol. 2021;101(11):adv00609. doi:10.2340/actadv.v101.565
9. Tribo MJ, Turroja M, Castano-Vinyals G, et al. Patients with moderate to severe psoriasis associate with higher risk of depression and anxiety symptoms: results of a multivariate study of 300 Spanish individuals with psoriasis. Acta Derm Venereol. 2019;99(4):417–422. doi:10.2340/00015555-3114
10. Malhotra SK, Mehta V. Role of stressful life events in induction or exacerbation of psoriasis and chronic urticaria. Indian J Dermatol Venereol Leprol. 2008;74(6):594–599. doi:10.4103/0378-6323.45100
11. Kamiya K, Kishimoto M, Sugai J, Komine M, Ohtsuki M. Risk factors for the development of psoriasis. Int J Mol Sci. 2019;20(18). doi:10.3390/ijms20184347
12. Mrowietz U, Dieckmann T, Gerdes S, Szymczak S, von Spreckelsen R, Körber A. ActiPso: definition of activity types for psoriatic disease: a novel marker for an advanced disease classification. J Eur Acad Dermatol Venereol. 2021;35(10):2027–2033. doi:10.1111/jdv.17434
13. Nast A, Altenburg A, Augustin M, et al. German S3-guideline on the treatment of psoriasis vulgaris, adapted from EuroGuiDerm - Part 1: treatment goals and treatment recommendations. J Dtsch Dermatol Ges. 2021;19(6):934. doi:10.1111/ddg.14508
14. Rendon A, Schakel K. Psoriasis pathogenesis and treatment. Int J Mol Sci. 2019;20(6). doi:10.3390/ijms20061475
15. Bissonnette R, Ho V, Langley RG. Safety of conventional systemic agents and biologic agents in the treatment of psoriasis. J Cutan Med Surg. 2009;13(Suppl 2):S67–76. doi:10.2310/7750.2009.00023
16. Koo J. A randomized, double-blind study comparing the efficacy, safety and optimal dose of two formulations of cyclosporin, Neoral and Sandimmun, in patients with severe psoriasis. OLP302 study group. Br J Dermatol. 1998;139(1):88–95. doi:10.1046/j.1365-2133.1998.02319.x
17. Armstrong AW, Puig L, Joshi A, et al. Comparison of biologics and oral treatments for plaque psoriasis: a meta-analysis. JAMA Dermatol. 2020;156(3):258–269. doi:10.1001/jamadermatol.2019.4029
18. Kamata M, Tada Y. Efficacy and Safety of biologics for psoriasis and psoriatic arthritis and their impact on comorbidities: a literature review. Int J Mol Sci. 2020;21(5). doi:10.3390/ijms21051690
19. Ruggiero A, Potestio L, Martora F, Villani A, Comune R, Megna M. Bimekizumab treatment in patients with moderate to severe plaque psoriasis: a drug safety evaluation. Expert Opin Drug Saf. 2023;22(5):355–362. doi:10.1080/14740338.2023.2218086
20. Papp KA, Langley RG, Sigurgeirsson B, et al. Efficacy and safety of secukinumab in the treatment of moderate-to-severe plaque psoriasis: a randomized, double-blind, placebo-controlled Phase II dose-ranging study. Br J Dermatol. 2013;168(2):412–421. doi:10.1111/bjd.12110
21. Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis--results of two Phase 3 trials. N Engl J Med. 2014;371(4):326–338. doi:10.1056/NEJMoa1314258
22. Radi G, Campanati A, Diotallevi F, Bianchelli T, Offidani A. Novel therapeutic approaches and targets for treatment of psoriasis. Curr Pharm Biotechnol. 2021;22(1):7–31. doi:10.2174/1389201021666200629150231
23. Colloca L. The placebo effect in pain therapies. Ann Rev Pharmacol Toxicol. 2019;59:191–211. doi:10.1146/annurev-pharmtox-010818-021542
24. Schedlowski M, Enck P, Rief W, Bingel U. Neuro-bio-behavioral mechanisms of placebo and nocebo responses: implications for clinical trials and clinical practice. Pharmacol Rev. 2015;67(3):697–730. doi:10.1124/pr.114.009423
25. Sondermann W, Reinboldt-Jockenhofer F, Dissemond J, Pfaar O, Bingel U, Schedlowski M. Effects of patients’ expectation in dermatology: evidence from experimental and clinical placebo studies and implications for dermatologic practice and research. Dermatology. 2021;1–15. doi:10.1159/000513445
26. Sonthalia S, Sahaya K, Arora R, et al. Nocebo effect in Dermatology. Indian J Dermatol Venereol Leprol. 2015;81(3):242–250. doi:10.4103/0378-6323.155573
27. Colloca L, Barsky AJ. Placebo and nocebo effects. N Engl J Med. 2020;382(6):554–561. doi:10.1056/NEJMra1907805
28. Schafer I, Oltrogge JH, Nestoriuc Y, et al. Expectations and prior experiences associated with adverse effects of COVID-19 vaccination. JAMA Network Open. 2023;6(3):e234732. doi:10.1001/jamanetworkopen.2023.4732
29. Wensing M, Elwyn G. Methods for incorporating patients’ views in health care. BMJ. 2003;326(7394):877–879. doi:10.1136/bmj.326.7394.877
30. Holsken S, Krefting F, Schedlowski M, Sondermann W. Expectation-induced enhancement of pain, itch and quality of life in psoriasis patients: study protocol of a randomised controlled trial. BMJ Open. 2021;11(9):e047099. doi:10.1136/bmjopen-2020-047099
31. Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349–357. doi:10.1093/intqhc/mzm042
32. O’Brien BC, Harris IB, Beckman TJ, Reed DA, Cook DA. Standards for reporting qualitative research: a synthesis of recommendations. Acad Med. 2014;89(9):1245–1251. doi:10.1097/ACM.0000000000000388
33. European Medicines Agency. Cosentyx: EPAR - Product Information. Available from: https://www.ema.europa.eu/en/documents/product-information/cosentyx-epar-product-information_en.pdf.
34. Saunders B, Sim J, Kingstone T, et al. Saturation in qualitative research: exploring its conceptualization and operationalization. Qual Quant. 2018;52(4):1893–1907. doi:10.1007/s11135-017-0574-8
35. maxqda.com. MAXQDA, software for qualitative data analysis; 2022.
36. Kuckartz U. Qualitative Inhaltsanalyse: Methoden, Praxis, Computerunterstützung. 4. überarbeitete Auflage [Qualitative content analysis: methods, practice, digital applications, 4th ed.] [Book in German]. Beltz Verlagsgruppe; 2018.
37. Sumpton D, Kelly A, Tunnicliffe DJ, et al. Patients’ perspectives and experience of psoriasis and psoriatic arthritis: a systematic review and thematic synthesis of qualitative studies. Arthritis Care Res. 2020;72(5):711–722. doi:10.1002/acr.23896
38. Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI)--a simple practical measure for routine clinical use. Clin Exp Dermatol. 1994;19(3):210–216. doi:10.1111/j.1365-2230.1994.tb01167.x
39. Miniszewska J, Juczynski Z, Ograczyk A, Zalewska A. Health-related quality of life in psoriasis: important role of personal resources. Acta Derm Venereol. 2013;93(5):551–556. doi:10.2340/00015555-1530
40. Larsen MH, Krogstad AL, Aas E, Moum T, Wahl AK. A telephone-based motivational interviewing intervention has positive effects on psoriasis severity and self-management: a randomized controlled trial. Br J Dermatol. 2014;171(6):1458–1469. doi:10.1111/bjd.13363
41. Eissing L, Radtke MA, Zander N, Augustin M. Barriers to guideline-compliant psoriasis care: analyses and concepts. J Eur Acad Dermatol Venereol. 2016;30(4):569–575. doi:10.1111/jdv.13452
42. Augustin M, Alvaro-Gracia JM, Bagot M, et al. A framework for improving the quality of care for people with psoriasis. J Eur Acad Dermatol Venereol. 2012;26(Suppl 4):1–16. doi:10.1111/j.1468-3083.2012.04576.x
43. Nestoriuc Y, von Blanckenburg P, Schuricht F, et al. Is it best to expect the worst? Influence of patients’ side-effect expectations on endocrine treatment outcome in a 2-year prospective clinical cohort study. Ann Oncol. 2016;27(10):1909–1915. doi:10.1093/annonc/mdw266
44. Michnevich T, Pan Y, Hendi A, Oechsle K, Stein A, Nestoriuc Y. Preventing adverse events of chemotherapy for gastrointestinal cancer by educating patients about the nocebo effect: a randomized-controlled trial. BMC Cancer. 2022;22(1):1008. doi:10.1186/s12885-022-10089-2
45. Nast A, Smith C, Spuls PI, et al. EuroGuiDerm Guideline on the systemic treatment of Psoriasis vulgaris - Part 1: treatment and monitoring recommendations. J Eur Acad Dermatol Venereol. 2020;34(11):2461–2498. doi:10.1111/jdv.16915
46. PsoNet e.V. FfrPiD. Globaler Bericht zur Schuppenflechte. Deutsche Übersetzung des Global Report on Psoriasis”; 2016.
47. Lawson A, Tan AC, Naylor J, Harris IA. Is retrospective assessment of health-related quality of life valid? BMC Musculoskelet Disord. 2020;21(1):415. doi:10.1186/s12891-020-03434-8
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
